What is AOD-9604?
The hGH Fragment Studied for Fat Metabolism and Weight Regulation
Disclaimer: Information provided is for research and educational purposes only. AOD-9604 is not approved by the FDA for therapeutic or cosmetic use.
Introduction:
AOD-9604 is a synthetic peptide fragment of human growth hormone (hGH 177–191) engineered to replicate hGH’s fat-metabolism effects without altering IGF-1 or glucose levels.¹ In preclinical research, it promoted lipolysis (fat breakdown) and reduced lipogenesis (fat storage) through receptor-independent mechanisms.² ³
AOD-9604 Fast Facts
- Class: Synthetic hGH fragment analog (Tyr-hGH 177–191)¹
- Intended purpose: Stimulate fat metabolism while avoiding hGH’s growth or glucose effects²
- Mechanism: Increases fat oxidation, enhances lipolysis, suppresses lipogenesis³
- Distinct from hGH: Does not activate the growth hormone receptor or raise IGF-1² ⁴
- Development stage: Completed Phase II trials; discontinued after lack of clinical efficacy⁶ ⁹
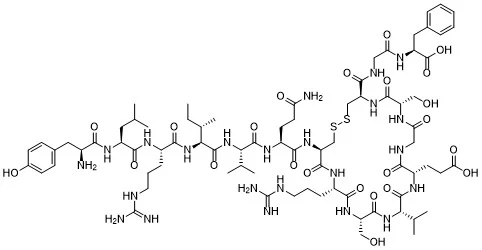
Chemical Structure
AOD-9604 is a 15-amino-acid fragment of human growth hormone. It was engineered isolate hGH’s lipolytic (fat-breaking) properties without activating the growth hormone receptor or increasing IGF-1.² The design allows it to mimic hGH’s metabolic domain in a shorter, more stable form.
How AOD-9604 Works (in Brief)
AOD-9604 was engineered from the C-terminal segment of human growth hormone (amino acids 177–191) — the portion thought to influence fat metabolism.¹ ²
In preclinical models, this fragment supported fat oxidation and lipid mobilization without the broader hormonal effects of full-length hGH.³ ⁴
Because it does not activate the traditional hGH receptor, its fat-metabolism effects are considered indirect and receptor-independent.²
Discovery & Research Milestones
Originally developed in the 1990s as a potential anti-obesity drug. While it showed anti-obesity signal⁶ and advanced into clinical trials, it failed to show sufficient weight-loss effects to support commercialization.⁸
| Year | Study & Source | Key Finding |
| 2000 2001 | Heffernan MA et al¹ ² ³ | AOD-9604 found to increase fat oxidation and reduce weight gain. |
| 2000 | Ng FM et al.⁴ | Demonstrated metabolic benefits without hGH-induced insulin resistance. |
| 2007 | Metabolic Pharmaceuticals⁷ | Reported 24-week Phase IIb results; discontinued anti-obesity program. |
| 2013 | Misra M ⁶ | 12-week trial showed modest signal; 24-week trial failed. Development stopped. |
| 2013 | Stier H et al. ⁵ | Summarized human safety data—well tolerated, no antibodies detected. |
AOD-9604 vs HGH Fragment 176–191: What’s the Difference?
Since AOD-9604 is often described as an HGH fragment, it’s easy to confuse the two. In reality:
- HGH Fragment 176–191 is the natural sequence of amino acids (176 to 191) at the C-terminal end of human growth hormone (HGH).
- AOD-9604 is a modified version of this fragment, designed to enhance stability and activity in fat metabolism. ¹²
- Takeaway: AOD-9604 should be thought of as an optimized, research-developed analog of HGH fragment 176–191
| Feature | HGH Fragment 176–191 | AOD-9604 |
| Origin | Direct amino acid sequence from HGH | Synthetic analog of fragment 176–191 |
| Primary Action | Stimulates lipolysis (fat breakdown) | Stimulates lipolysis + inhibits lipogenesis |
| Stability | Shorter half-life, limited clinical exploration | Designed to be more stable and suitable for trials |
| Clinical Development | Investigated mainly in preclinical studies | Entered Phase I/II obesity trials |
Takeaway
AOD-9604 should be thought of as an optimized, research-developed analog of HGH fragment 176–191 — not an entirely separate peptide, but not exactly the same either. This distinction helps explain why it progressed into formal obesity trials, whereas the raw fragment itself did not.
Regulatory Status
AOD-9604 is not FDA-approved for any medical use and is listed by the FDA as a bulk drug substance that may present safety risks.⁸ Clinical development was halted after Phase IIb trials failed to meet efficacy endpoints.⁹
Summary
AOD-9604 is a synthetic analog of the HGH 177–191 fragment developed for fat-metabolism research. Preclinical studies showed lipolytic and anti-lipogenic effects, but human trials failed to demonstrate significant weight-loss outcomes. It remains available for research use only.
FAQs About AOD-9604
What is AOD-9604?
AOD-9604 is a synthetic analog of the HGH fragment 176–191, studied for fat metabolism and weight regulation.
Is AOD-9604 the same as HGH fragment 176–191?
Not exactly — AOD-9604 is a modified version of the natural fragment, designed to be more stable and active.
Does AOD-9604 build muscle like HGH?
No, it does not stimulate IGF-1 or muscle growth — its effects are limited to fat metabolism pathways.
Is AOD-9604 approved for medical use?
No, AOD-9604 is not FDA-approved and is available only for research purposes.
Related Articles
- How Does AOD-9604 Work
- AOD-9604 Benefits
- AOD-9604 Side Effects & Safety
- What is Ipamorelin
- What is BPC-157
References
- Heffernan MA, et al. Effects of oral administration of a synthetic fragment of human growth hormone on lipid metabolism in rodents and in human adipose tissue. Am J Physiol Endocrinol Metab. https://pubmed.ncbi.nlm.nih.gov/10950816/
- Heffernan MA, et al. Increase of fat oxidation and weight loss in obese mice by a fragment of human growth hormone. Obes Res. 2001;9(5):341–347. https://pubmed.ncbi.nlm.nih.gov/11673763/
- Heffernan M, et al. The effects of human GH and its lipolytic fragment (AOD9604) on lipid metabolism in mice. Endocrinology. 2001;142(12):5182–5189. https://pubmed.ncbi.nlm.nih.gov/11713213/
- Ng FM, et al. Metabolic studies of a synthetic lipolytic domain (AOD9604) of human growth hormone in vitro and in vivo. Ann N Y Acad Sci. 2000;921:262–272. https://pubmed.ncbi.nlm.nih.gov/11146367/
- Stier H, et al. Safety and tolerability of the hexadecapeptide AOD9604 in humans. J Endocrinol Metab. 2013;3(1–2):7–15. https://jofem.org/index.php/jofem/article/view/157
- Misra M. Obesity pharmacotherapy: current perspectives and future directions. Curr Cardiol Rev. 2013;9(1):33–54. https://pmc.ncbi.nlm.nih.gov/articles/PMC3584306/
- Cox HD, et al. Detection and in vitro metabolism of AOD9604. Drug Test Anal. 2015;7(9):808–814. https://pubmed.ncbi.nlm.nih.gov/25208511/
- Metabolic Pharmaceuticals (ASX announcement). Phase 2B results for AOD9604 do not support commercial viability (obesity indication). Feb 21, 2007. https://www.asx.com.au/asxpdf/20070221/pdf/3111t0ww55jr72.pdf