What is Melanotan-II (MT-2)?
The melanocortin peptide studied for tanning and sexual function
Disclaimer: Information provided is for research and educational purposes only. Melanotan-II is not approved by the FDA or EMA for human or cosmetic use.
Introduction:
Melanotan-II (MT-II), often called the “tanning peptide,” is a synthetic analog of the natural α-melanocyte-stimulating hormone (α-MSH). It was developed in the 1980s to explore sunless tanning and skin-protection effects.¹ As a melanocortin receptor agonist, Melanotan-II promotes skin pigmentation and has been studied for its influence on sexual function, energy balance, and UV protection.¹ ² ³ ⁴ ⁸ ⁹
MT-II Fast Facts
- Class: Melanocortin receptor agonist (α-MSH analog)
- Design: Cyclic heptapeptide (lactam bridge) engineered for potency/stability
- Primary targets: MC1R (pigmentation), central MC3R/MC4R (sexual function, energy balance).⁸
- Most studied effects: Increased pigmentation; erectogenic response in ED cohorts; nausea/yawning common.¹
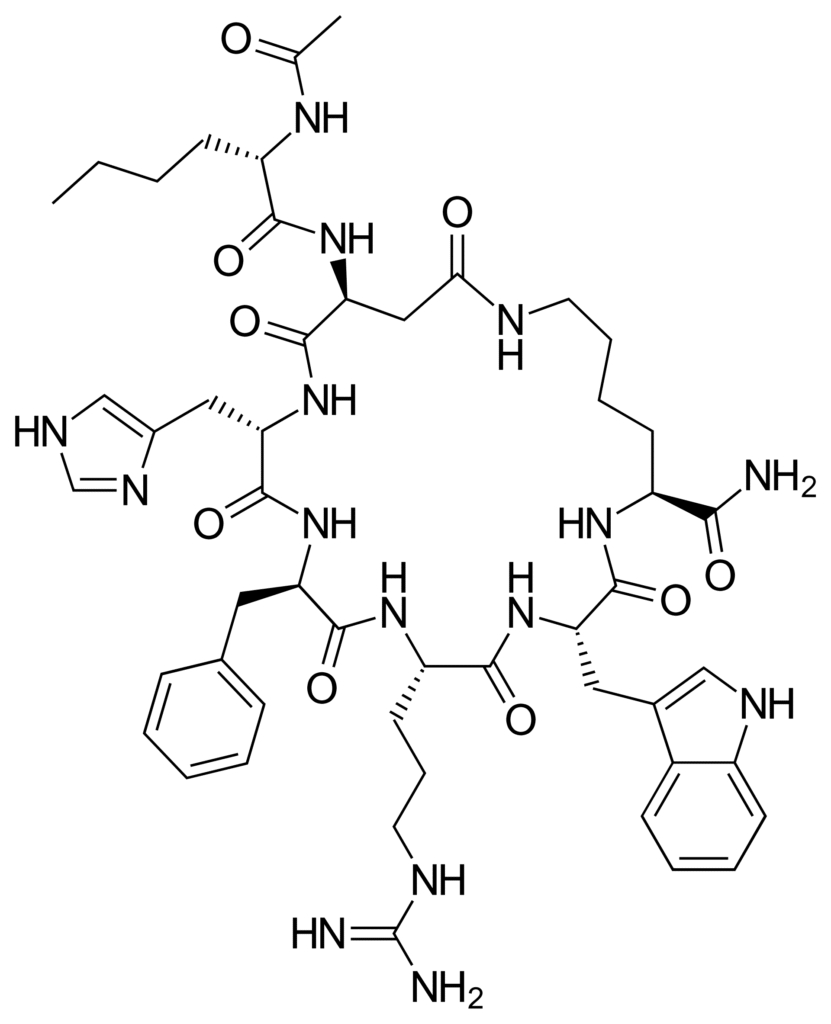
Melanotan-2 Chemical Structure
What exactly does MT-II do?
MT-II stimulates melanogenesis via MC1R on melanocytes → darker pigmentation following short subcutaneous dosing.¹
In the CNS, MT-II can trigger erections without sexual stimulation, pointing to central melanocortin circuitry (hypothalamic and spinal sites).²
Discovery & Research Milestones
- Originated at the University of Arizona in the 1980s, where researchers were exploring sunless tanning agents.¹
- Built as a short, stable analog of α-MSH, improving its half-life and activity compared to the natural hormone.²
- Early studies tested Melanotan II in skin pigmentation disorders and sexual dysfunction, but it never achieved regulatory approval.³
| Year | Study & Source | Key Finding |
| 1996 | Dorr RT et al. | Visible tanning after low-dose MT-II; common side effects include nausea & yawning. Recommended 0.025 mg/kg. |
| 1998 | Wessells H et al. | First studies on use for erectile dysfunction. MT-2 induces erections without erotic stimuli |
| 2000-2003 | Wessells H et al. | Mechanism established: central melanocortin pathways (hypothalamus and spinal cord) mediate MT-II-induced erection. |
| 2003-2004 | PT-141 program | MT-II signal translated into PT-141/bremelanotide; early RCTs show dose-dependent erectile activity and acceptable tolerance. |
A Closer Look: What the Research Shows
- Skin Pigmentation (Tanning)
A single-blind, alternating-day phase-I trial found visible tanning after five low doses and recommended 0.025 mg/kg for future studies; common AEs were nausea, yawning, somnolence. - Erectile physiology
A double-blind, placebo-controlled crossover study showed robust erections (average of 38 min of >80% rigidity) with 0.025 mg/kg MT-II; nausea/yawning increased vs. placebo. - Development of PT-141
The erectogenic signal from MT-II led to development of PT-141/bremelanotide (a related melanocortin agonist)
Research Applications
- Pigmentation disorders: Investigated for use in vitiligo and erythropoietic protoporphyria.²
- Skin protection: Studied for potential reduction of UV-related skin damage.³
- Sexual function: Explored as a treatment for erectile dysfunction and female sexual arousal disorder. ³
Regulatory Status
- MT-II: Not approved for tanning or sexual function
- Afamelanotide (MT-I) is approved for EPP (EU 2014; US 2019); long-term safety/PK are reviewed separately.
Summary
Melanotan-2 is a potent melanocortin agonist that darkens skin (via MC1R) and triggers erections by acting on central melanocortin pathways. Controlled early trials defined doses and common AEs; the erectogenic signal catalyzed development of bremelanotide.
FAQs About Melanotan II
Is Melanotan II FDA-approved?
No, it is not approved by the FDA or EMA and is considered a research chemical.
What does Melanotan II do?
It increases melanin production in the skin by activating melanocortin receptors, leading to darker pigmentation.
What was Melanotan II originally developed for?
It was developed as a tanning agent and studied for pigmentation disorders and sexual dysfunction.
Related Articles
Related Products
Related Articles
References
- Dorr RT, Lines R, Levine N, et al. Evaluation of melanotan-II, a superpotent cyclic melanotropic peptide in a pilot phase I clinical study. Life Sci. 1996;58(20):1777-1784. https://pubmed.ncbi.nlm.nih.gov/8637402/
- Wessells H, Fuciarelli K, Hansen J, et al. Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction: double-blind, placebo-controlled crossover study. J Urol. 1998;160(2):389-393. https://pubmed.ncbi.nlm.nih.gov/9679884/
- Vemulapalli R, Kurosawa M, Killinger J, et al. Activation of central melanocortin receptors by melanotan-II (Ac-Nle-c[Asp-His-D-Phe-Arg-Trp-Lys]-NH₂) results in penile erection in rats. Neuroscience. 2001;106(3):547-552. https://pubmed.ncbi.nlm.nih.gov/11591452/
- Wessells H, Hruby V J, Hackett J, et al. Central melanocortin pathways mediate the erectogenic effects of melanotan-II in animal models. J Urol. 2003;170(6 Pt 1):S. (Abstract and mechanistic report.) https://pubmed.ncbi.nlm.nih.gov/14665870/
- Rosen R C, Diamond L E, Earle D C, Shadiack A M, Molinoff P B. Evaluation of the safety, pharmacokinetics and pharmacodynamics of intranasal PT-141, a melanocortin receptor agonist, in healthy males and patients with erectile dysfunction. J Sex Med. 2004;1(3):193-202. https://pubmed.ncbi.nlm.nih.gov/14999221/
- Diamond L E, Earle D C, Garcia W D, Spana C. Double-blind, placebo-controlled evaluation of the safety, efficacy and dose response of subcutaneous PT-141 in men with erectile dysfunction. Int J Impot Res. 2004;16(4):353-360. https://pubmed.ncbi.nlm.nih.gov/14963471/
- Hadley M E. Discovery that a melanocortin regulates sexual functions in male and female humans. Peptides. 2005;26(10):1687-1691. https://pubmed.ncbi.nlm.nih.gov/15996790/
- Cai M, Hruby V J. Melanocortin receptors: their structure, function, and ligands. Peptides. 2016;88:3-18. https://pubmed.ncbi.nlm.nih.gov/27746082/ (Open access PMC article: https://pmc.ncbi.nlm.nih.gov/articles/PMC5999398/)
- Mun Y, Jeon J H, Lee E S, et al. Melanocortin-1 receptor (MC1R): pharmacology and therapeutic prospects. Pharmaceutics. 2023;15(7):1814. https://pmc.ncbi.nlm.nih.gov/articles/PMC10418475/
- Hjuler K F, Brøgger-Mikkelsen M, Pedersen S, et al. Melanoma associated with the use of melanotan-II. Acta Derm Venereol. 2014;94(1):115-116. https://pubmed.ncbi.nlm.nih.gov/24355990/
- DermNet NZ. Melanotan II – Uses and side effects. (Clinical dermatology resource.) https://dermnetnz.org/topics/melanotan-ii
- Minder E I, Schneider-Yaniv L, Schneider-Yaniv Z. Pharmacokinetics and pharmacodynamics of afamelanotide, a melanocortin 1 receptor agonist, in humans. Clin Pharmacokinet. 2017;56(8):815-826. https://pubmed.ncbi.nlm.nih.gov/28063031/