What Is TB-500?
The Synthetic Peptide Modeled After Thymosin Beta-4 for Soft-Tissue Repair
Disclaimer: Information provided is for research and educational purposes only. TB-500 is not approved by the FDA or any regulatory agency for human or veterinary use.
Introduction: What Is TB-500?
TB-500 (sometimes written as TB500) is a synthetic peptide modeled after a key segment of thymosin beta-4 (Tβ4), a naturally occurring protein found in almost all animal tissues. It has been studied for its role in tissue repair and regeneration, especially in muscle, tendon, ligament, and wound-healing models.¹ ²
Preclinical research demonstrates that TB-500 promotes cell migration, angiogenesis, and tissue repair—making it a unique compound among regenerative peptides².
TB-500 At-a-Glance
- Type: Synthetic peptide fragment (N-terminal region of thymosin beta-4)
- Sequence: Ac-SDKP or longer fragments (e.g., LKKTETQ)
- Discovered: Late 1990s–early 2000s, building on Tβ4 research from the 1960s–1990s
- Key Features: Binds actin, stimulates angiogenesis and cell migration
- Primary Research: Animal models of muscle, tendon, ligament, cardiac, and wound healing
Chemical Structure & Origin
TB-500’s peptide sequence—typically Ac-SDKP or longer N-terminal fragments—confers its strong actin-binding and cell migration effects. As a synthetic analog, TB-500 is not found naturally but is based on a conserved active region within the Tβ4 peptide.
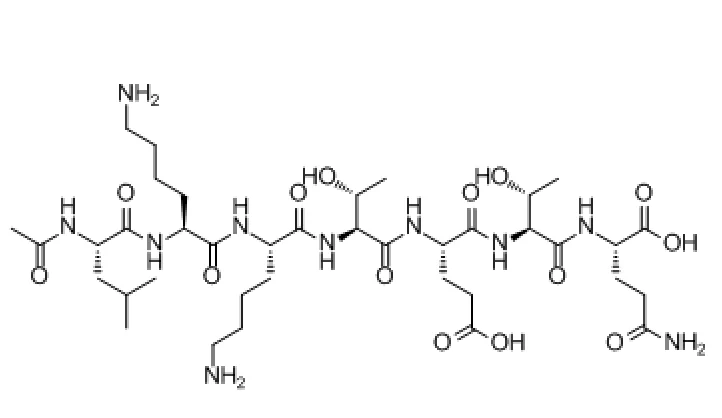
TB-500 peptide structure and amino acid sequence
How TB-500 Works (in Brief)
TB-500 exerts its effects through pathways shared with thymosin β-4, its parent peptide. It binds to actin, a major cellular structural protein, promoting cell migration, angiogenesis, and tissue remodeling in response to injury.³⁴ By facilitating the movement of repair cells and supporting new blood-vessel growth, TB-500 contributes to accelerated healing in preclinical models.²
Discovery and Key Research Milestones
The history of TB-500 is rooted in the discovery of thymosin beta-4, a 43-amino-acid peptide isolated in the 1960s–1970s from the thymus gland. Scientists later identified a specific sequence—responsible for most of Tβ4’s actin-binding and healing properties—and synthesized it as TB-500 for research use³. Unlike full-length Tβ4, TB-500 is optimized for laboratory synthesis, stability, and cost-effectiveness.
| Year | Study & Source | Key Finding |
| 1977 | Goldstein AL et al., PNAS (PMID 265536)⁴ | Thymosin fractions isolated from thymus |
| 1997 | Malinda KM et al., FASEB J (PMID 9194528)² | Tβ4 (and by analogy TB-500) promotes cell migration, angiogenesis |
| 1996/ 1999 | Van Troys et al., EMBO J (PMID 8617195)⁵; Safer et al., Biochemistry (PMID 9153421)⁶ | Tβ4 actin-binding site mapped; actin-sequestration mechanism clarified |
| 2002 | Huff T et al., FEBS Lett (PMID 11978733)⁷ | Tβ4 released from platelets and cross-linked to fibrin/collagen; relevance to wound healing |
| 2005 | Goldstein AL et al., Trends Mol Med (PMID 16099219)³ | Tβ4 “moonlights” as both actin-sequestering and tissue repair peptide. |
Note: Nearly all data is from animal or in vitro models; human clinical trials are rare and preliminary.
How TB-500 Differs From Other Peptides
Related Compounds at a Glance
| Peptide | Main Use | Oral? | Key Difference |
TB-500 | Soft tissue repair | Yes | Targets actin, cell migration |
BPC-157 | Multi-tissue repair | Yes | Stable in GI tract; broad activity |
| GHK-Cu | Skin & cosmetic | No | Copper-binding; collagen stimulation |
Conclusion & Key Takeaways
TB-500 is a synthetic peptide fragment designed to harness the regenerative properties of thymosin beta-4. Its ability to stimulate cell migration and new blood vessel formation has made it a popular research tool for studies of muscle, tendon, ligament, and wound healing—especially in animal and veterinary contexts.
FAQs About TB-500
What is TB-500?
TB-500, also written TB 500, is a synthetic peptide fragment derived from thymosin beta-4. It is studied for its healing and regenerative effects in muscle, tendon, and wound models.
Is TB-500 the same as thymosin beta-4?
No. TB-500 is a short synthetic version of part of thymosin beta-4, not the full-length natural peptide.
Why is TB-500 popular in research?
Researchers study TB-500 for its ability to promote cell migration, blood vessel growth, and healing in animal models of muscle, tendon, and wound injury.
Related Articles
- How Does TB-500 Work? Mechanism of Action
- TB-500 Benefits
- TB-500 Safety & Side Effects
- TB-500 vs. BPC-157: Comparison
- What is BPC-157?
- What is GHK-CU?
References
- Huff T, Müller CS, Otto AM, Netzker R, Hannappel E. β-Thymosins, small acidic peptides with actin-binding properties. FEBS Letters. 2001;528(1-3):27–33. https://www.sciencedirect.com/science/article/abs/pii/S135727250000087X
- Malinda KM, Goldstein AL, Kleinman HK. Thymosin β4 stimulates directional migration of human umbilical vein endothelial cells. FASEB J. 1997;11(7):474–481.https://pubmed.ncbi.nlm.nih.gov/9194528/
- Goldstein AL, Hannappel E, Kleinman HK. Thymosin beta 4: actin-sequestering protein moonlights to repair injured tissues. Trends Mol Med. 2005;11(9):421-429. https://pubmed.ncbi.nlm.nih.gov/16099219/
- Goldstein AL, Guha A, Zatz MM, Hardy MA, White A. Purification and biological activity of thymosin, a hormone of the thymus gland. Proc Natl Acad Sci USA. 1972;69(7):1800–1803. [PMID: 4505666]
https://pubmed.ncbi.nlm.nih.gov/4505657/ - Safer D, Elzinga M, Nachmias VT. Thymosin β4 and Fx, an actin-sequestering peptide, are indistinguishable. J Biol Chem. 1991;266(7):4029–4032. [PMID: 1995622] https://pubmed.ncbi.nlm.nih.gov/1999398/
- Van Troys M, Vandekerckhove J, Ampe C. Structural modules in actin-binding proteins: towards a new classification. FEBS Lett. 1999;399(1-2):1–7. [PMID: 8617195] https://pubmed.ncbi.nlm.nih.gov/9990286/
- Huff T, Müller CS, Otto AM, Netzker R, Hannappel E. Thymosin β4 is released from human blood platelets and attached to fibrin and collagen. FASEB J. 2002;16(6):691–696. [PMID: 11978733] https://pubmed.ncbi.nlm.nih.gov/11978733/