How to Read a Peptide COA (Real vs Misleading)
Understanding and interpreting a Certificate of Analysis (COA): A Researcher’s Field Guide
Disclaimer: All information presented in this article is strictly for scientific, academic, and educational purposes. Research peptides discussed here are intended solely for laboratory research and in vitro studies. They are not approved by the FDA or any regulatory agency for human or veterinary use, clinical applications, therapeutic use, or consumption of any kind.
1. Introduction
A Certificate of Analysis (COA) is the single most important document in the research peptide supply chain. It transforms an unlabeled white powder into a scientifically verifiable reagent.
For Research Use Only (RUO) peptides—materials that are not approved for human or veterinary use—the COA is your primary quality assurance tool. Because RUO peptides are not regulated like pharmaceuticals, the burden of verification sits with the researcher. A legitimate COA must answer four core questions:
- Identity – Is this the correct sequence? (Mass Spectrometry)
- Purity – Is it chemically clean? (HPLC)
- Content – How much actual peptide is present? (Net Peptide Content)
- Contaminants – Are there any harmful compounds also present? (Heavy Metals, Endotoxins)
Credible suppliers use independent, third-party laboratories to generate COAs. Third-party testing eliminates conflict of interest and gives the researcher unbiased, empirical data—not marketing claims.
Note: Most 3rd Party providers group identity, purity and content into a single test result (e.g., Janoshik, annotated below). Contaminants usually require separate tests – typically 1 test for endotoxins and 1 for heavy metals.

2. HPLC Purity — The “Cleanliness” Check
High-Performance Liquid Chromatography (HPLC) is the standard method for quantifying peptide purity. It separates the target peptide from peptide-related impurities (deletion sequences, truncated fragments, and synthesis by-products).
What the Purity Percentage Actually Means
The COA will list a purity value such as 98.7%. This represents:
(Area of the target peak) ÷ (Total area of all UV-absorbing peptide-related peaks)
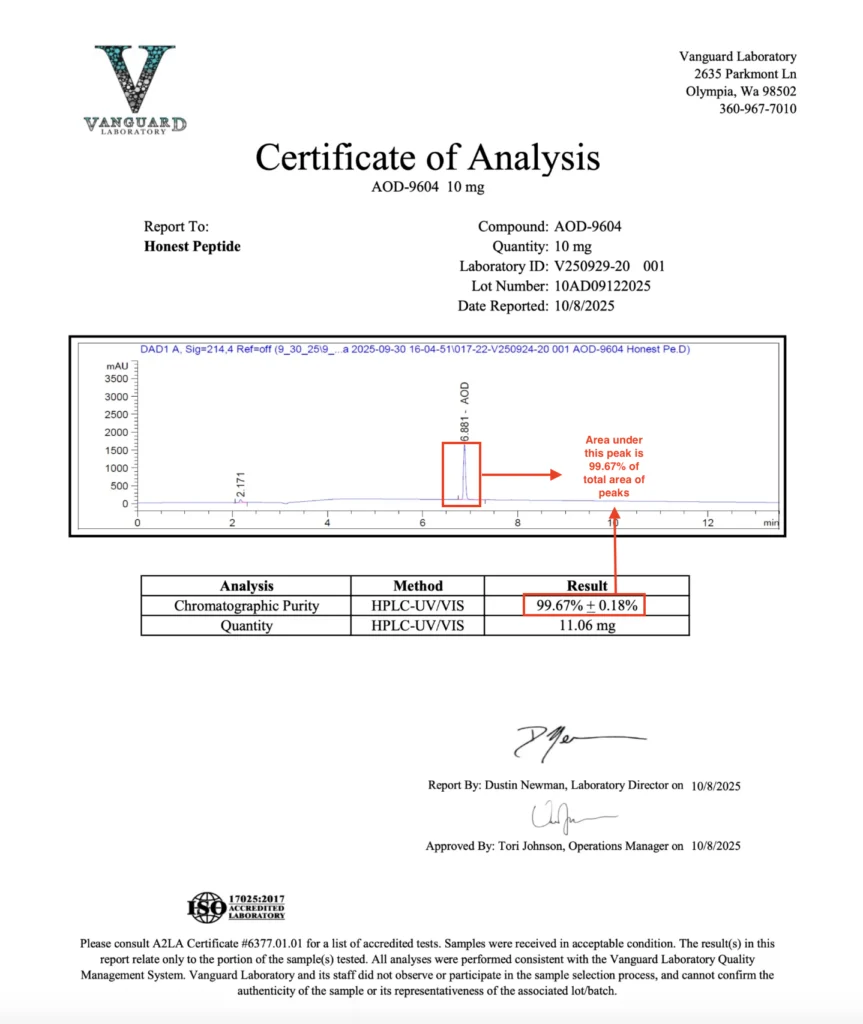
Important nuance:
HPLC purity reflects only UV-absorbing organic impurities, not water, salts, or non-chromophore contaminants. This is why purity and Net Peptide Content differ.
How to Interpret It
- ≥99% — Excellent. Ideal for quantitative assays, structural biology, binding studies, or sensitive cell work.
- 95–98% — Standard RUO grade. Suitable for most discovery-phase research.
- <95% — Potentially problematic for precision assays.
Chromatogram or No Chromatogram?
Some labs (e.g., Janoshik) provide numerical purity reports without chromatogram images. Others attach full chromatograms. A lack of chromatogram does not invalidate the results, rather it is a function of how they present results to customers. However, as a best practice we recommend asking a new tester for the chromatogram file to ensure it matches what they have provided in the test result.
3. Mass Spectrometry — The “Identity” Check
HPLC cannot confirm sequence identity. A peptide missing one amino acid may appear 99% pure on HPLC, but would be 0% the desired peptide.
Mass Spectrometry (MS) is the only reliable way to confirm identity.
Most 3rd party testers provide both MS & HPLC in a single test and will only alert you in the event that Mass Spec does not detect the correct sequence. As a best practice, always confirm that your tester is providing MS.
4. Net Peptide Content — The “Missing Mass”
This is the most misunderstood metric in peptide QC. And unfortunately no tester does this well.
Purity vs. Net Content
- Purity = peptide content vs. peptide impurities
- Net Peptide Content = peptide vs. everything else (water + salts + counter-ions)
A peptide can be 99% pure yet 70–85% net content. This is because HPLC only captures peptide related content, and therefore the 1% residual is composed only of peptide-related impurities. It fails to capture anything non-peptide related.
Why Net Content Is Always <100%
Lyophilized peptides bind mutiple forms of non-peptide content, including:
- Residual moisture (hygroscopic behavior, especially Lys/Arg-rich sequences)
- Counter-ions (TFA, Acetate, HCl)
- Atmospheric water during freeze-drying transfer
Practical Impact
If you dissolve a 5 mg vial assuming all 5 mg is active peptide, your molar concentration will be incorrect.
Example:
- Gross weight: 5 mg
- Net content: 80%
- Actual peptide mass: 4 mg
Always adjust dosing based on net content when performing precise quantitative work.
Where Testers Fall Short
Most testers provide net peptide content in their results. However, they do not provide gross peptide content. As a result it is impossible to calculate the gross-to-net ratio (net weight/gross weight) and thereby not possible to ascertain how much non-peptide content is contained in the product
This is mostly harmless as most residue (TFA, residual moisture) is benign. However, there is harmful non-peptide content, which must be screened for. This is where contaminant testing comes into play.
5. Contaminant Testing — Filtering out Harmful Compounds
When evaluating peptide quality, most people focus on purity (HPLC) and identity (mass spec). Those confirm you have the correct molecule and that it’s relatively clean from peptide-related byproducts.
However, there is a separate and equally important category: Non-peptide contaminants
Two of the most important are heavy metals and endotoxins.
Heavy Metals
Heavy metals are trace inorganic elements that can enter the product during:
- Raw material sourcing
- Manufacturing equipment contact
- Poorly controlled synthesis environments
The primary metals typically screened are Lead (Pb), Cadmium (Cd), Mercury (Hg), Arsenic (As).
Heavy metals are invisible to HPLC and mass spec and, if present, will accumulate in organic matter offer time with negative effects on neurological, renal and hepatic function.
It is therefore crucial to test for heavy metals; accordingly, Honest Peptide tests for every single product for heavy metals.
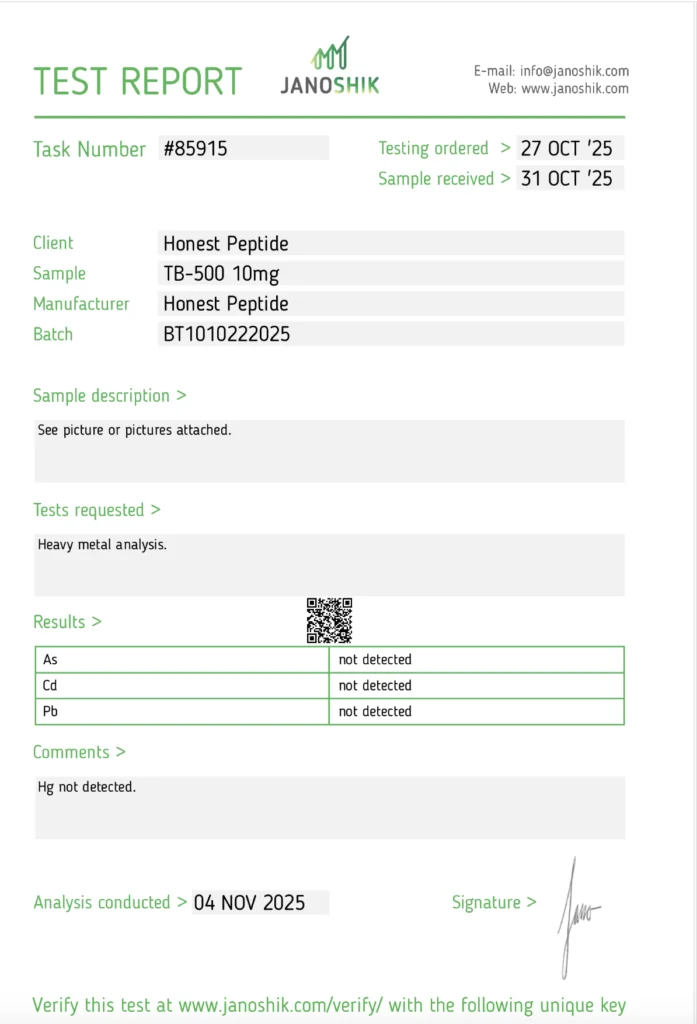
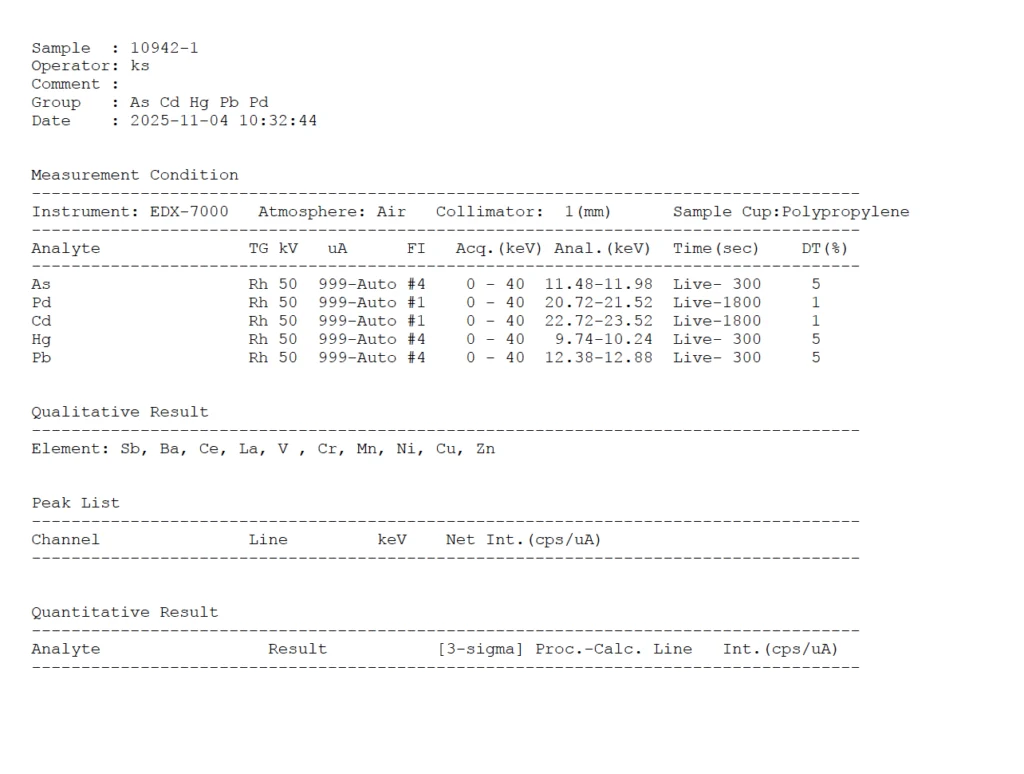
The standard method for heavy metals testing is ICP-MS (Inductively Coupled Plasma Mass Spectrometry), which detects metals as parts-per-billion (ppb) levels. The primary metals screened are Lead (Pb), Cadmium (Cd) and Arsenic (As)
The test output is typically delivered as a binary detected or not detected, but a legitimate tester should also be able to produce metal-by-metal quantitative detail upon request.
Summary Results (Janoshik)
Detailed Results (Janoshik) – UPON REQUEST
Endotoxins
Endotoxins are Lipopolysaccharides (LPS) from the outer membrane of Gram-negative bacteria. They are not “live bacteria” but fragments of bacterial cell walls. They can enter peptides if:
- Water systems are not properly controlled
- Manufacturing environments lack sterility
- Filtration steps are inadequate
As they are not live bacteria, endotoxins will not cause any kind of infection. However, sustained exposure can trigger immune reactions leading to fever and inflammation.
The standard method for endotoxin testing is LAL Test (Limulus Amebocyte Lysate assay). This assay detects endotoxin activity at very low levels and reports results in EU/ml (Endotoxin Units per milliliter) or in some cases EU/vial.
Some 3rd party labs provide the results of the assay as a binary pass/fail. Others provide an exact EU number.
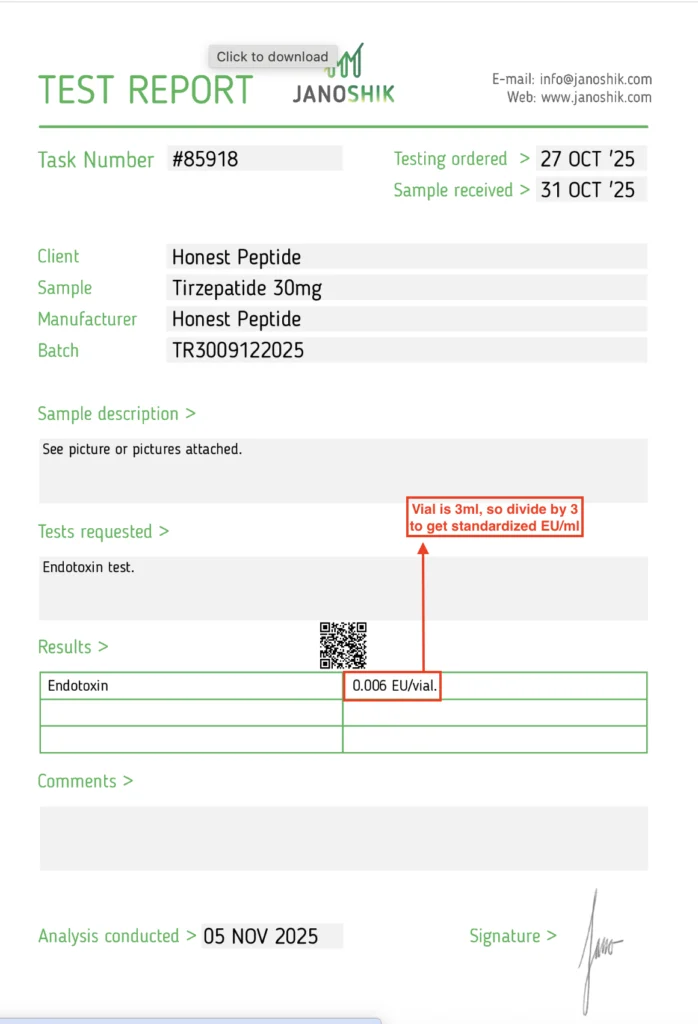
Example of Binary Pass/Fail Test

5. How to Evaluate Claims — The “Trust But Verify” Model
In RUO peptides, data is everything. A static PDF alone is not enough.
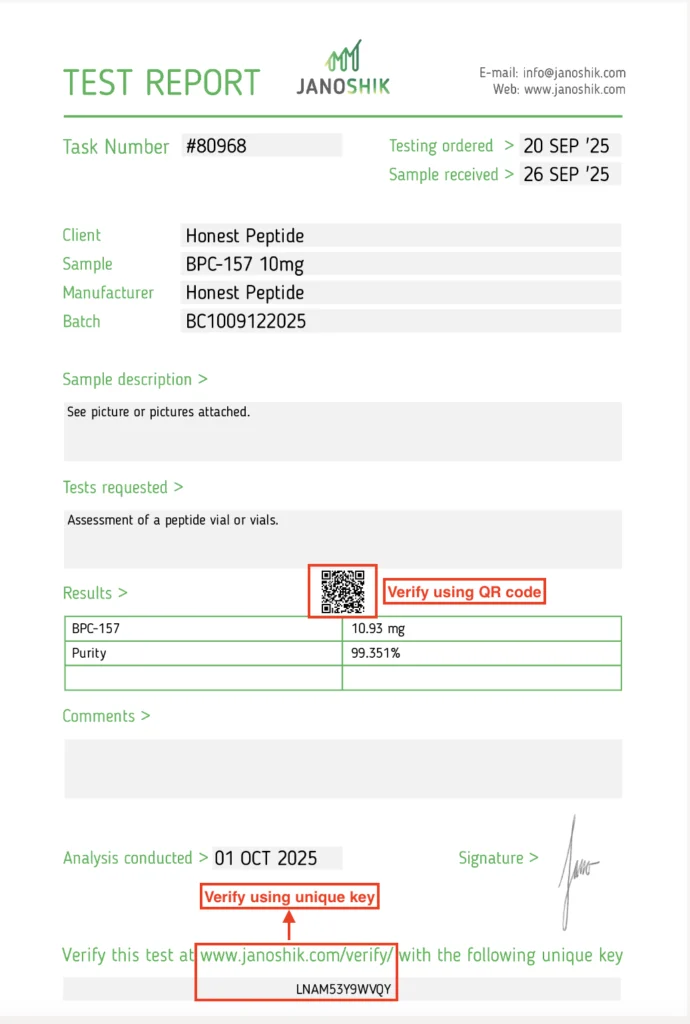
1. Digital Verification (Required)
Top-tier analytical labs provide:
- Verification Keys
- COA IDs
- QR codes
- Database lookup portals
A legitimate COA must be verifiable directly on the lab’s server. Further, a serious researcher must verify the COA on the 3rd party testers website. Otherwise, it is very easy for unscrupulous suppliers to photoshop results. Indeed, we see this all the time
2. Technical Transparency
A credible vendor is clear about:
- Salt form (TFA vs Acetate)
- Net peptide content
- Purity measurement method
- Identity verification method
- Where the testing was performed
Opaque suppliers should be treated with caution.
6. RUO vs. GMP — Managing Expectations Accurately
A COA for a Research Use Only peptide is not:
- A sterility certificate
- A pyrogen (endotoxin) certificate
- A GMP batch record
- Evidence of suitability for human use
- FDA-reviewed documentation
RUO COAs Contain:
- Sequence identity (MS)
- Chemical purity (HPLC)
- Net content
- Physical description
RUO COAs Do Not Contain:
- Sterility testing (unless test is added)
- Endotoxin testing (unless test is added)
- Bioburden testing
- Viral or microbial safety data
- GMP compliance statements
- Clinical suitability assessments
7. What a COA Cannot Tell You (Important for Research Planning)
A COA cannot determine:
- Whether your peptide is sterile
- Whether the material is free of pyrogens
- How the peptide should behave biologically
- Whether impurities are biologically benign
- How stable the peptide will be after reconstitution
- Whether the vendor followed GMP manufacturing controls
These limitations apply to the entire RUO category, not just a specific supplier.
8. Summary
A peptide COA is your primary quality assurance document.
To evaluate a peptide, confirm:
- HPLC Purity — How clean is it?
- Mass Spec Identity — Is the sequence correct?
- Net Peptide Content — How much peptide is actually present?
- Non Peptide Contaminants – Heavy Metals & Endotoxins
- Digital Verification – Always cross-check COAs on 3rd Party lab website
9. Related Articles
Explore the rest of our other Pillar Pieces in our Research Hub:
- What are Research Peptides? [LINK HERE]
Learn what research peptides are under the FDA RUO framework, how they differ from GMP/clinical material, and what that means for legality, QC, and lab use.
- The Complete Guide to Research Peptides (RUO) [LINK HERE]
RUO research peptide reference covering RUO framework, manufacturing, HPLC/MS testing, COAs, storage and vendor evaluation in one definitive guide.
- How Are Peptides Made? [LINK HERE]
Step-by-step guide to how peptides are made: SPPS, cleavage, HPLC purification, lyophilization, and QC so you can interpret COAs and compare RUO suppliers.
- Definitive Guide to Peptide Storage & Stability [LINK HERE]
Practical SOP for peptide storage, reconstitution, and aliquoting. Understand degradation pathways and keep RUO peptides stable and sterile.
- How to Select a Peptide Vendor [LINK HERE]
Tips on how to select a peptide vendor: due diligence checklist for RUO peptides, transparency, COA quality, cold chain, pricing, and marketing red flags.
10. FAQs
How do I verify a third-party COA?
Use the verification key, QR code, or database lookup. If unsure, email the testing lab with the Report ID.
Why is there no batch number on my vial?
RUO products often omit vial-level batch IDs. The COA itself is the batch record.
How do I verify a third-party COA?
Use the verification key, QR code, or database lookup. If unsure, email the testing lab with the Report ID.
What should I do if purity is <98%?
For exploratory work it may be acceptable, but for quantitative assays impurities can skew results. If the purity is lower than advertised, contact the vendor.
Does the COA guarantee endotoxins or sterility testing?
No. RUO COAs do not include endotoxin or sterility testing, although these are occasionally added on by RUO vendors.
References
- Bachem. “Quality Control of Amino Acids & Peptides: A Guide.” 2025.
- Numan, S. “Certificates of Analysis for Peptides: What Researchers Need to Know.” Verified Peptides, 2025.
- United States Pharmacopeia (USP). General Chapter <1043>. 2021.